what is an fda prestar?
Published Streamed 1 year ago • 560 plays • Length 21:42Download video MP4
Download video MP3
Similar videos
-
 4:34
4:34
what is an fda pre-submission?
-
 18:21
18:21
how does the fda prestar impact the fda submission process and new product development?
-
 12:06
12:06
what is a class 1 and 2 device exemption?
-
 23:24
23:24
what is an fda us agent? and why do you need one?
-
 2:34
2:34
what is an fda prestar?
-
 54:27
54:27
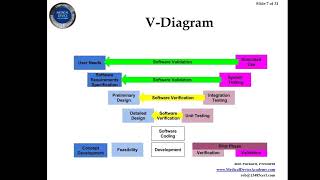
software validation documentation for medical devices - fda estar
-
 1:06:28
1:06:28
mastering your 510(k) submission process
-
 1:15:28
1:15:28
how to you create a design history file (dhf)?
-
 1:48
1:48
what is an fda class 1 medical device? - countyoffice.org
-
 18:53
18:53
what is the regulatory pathway for a de novo medical device or ivd?
-
 2:19
2:19
understanding the fda medical device 510k process
-
 17:20
17:20
fda estar - general info - #1 in the 510(k) estar series
-
 12:59
12:59
what is a medical device? understanding the fda definition and key differences from drugs
-
 15:35
15:35
what is a de novo?
-
 5:22
5:22
what does an fda us agent do?
-
 7:30
7:30
what is the software precertification (pre-cert) pilot program?
-
 14:42
14:42
what is the best approach to private labeling medical devices?
-
 22:34
22:34
fda establishment registration and listing for medical devices
-
 16:38
16:38
do you know the difference between a medical device component and an accessory medical device?
-
 2:45
2:45
changes to the fda ecopy submission process
-
 4:11
4:11
what is the difference between a 510k and de novo?