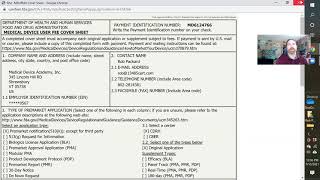
what is an fda us agent? and why do you need one?
Published Streamed 1 year ago • 867 plays • Length 23:24Download video MP4
Download video MP3
Similar videos
-
 5:22
5:22
what does an fda us agent do?
-
 6:11
6:11
is your fda us agent a consulting resource?
-
 21:42
21:42
what is an fda prestar?
-
 12:06
12:06
what is a class 1 and 2 device exemption?
-
 4:25
4:25
understanding the difference between fda approval and fda clearance
-
 14:42
14:42
what is mdsap?
-
 54:27
54:27
software validation documentation for medical devices - fda estar
-
![regulatory pathway: reach the us market [class ii 510k exempt]](https://i.ytimg.com/vi/BWK7LQPt8ZY/mqdefault.jpg) 32:50
32:50
regulatory pathway: reach the us market [class ii 510k exempt]
-
 12:59
12:59
what is a medical device? understanding the fda definition and key differences from drugs
-
 7:29
7:29
family health insurance deductible explained - embedded vs. aggregate. it's confusing.
-
 20:05
20:05
after successful fda approval, what do you need to do next?
-
 22:34
22:34
fda establishment registration and listing for medical devices
-
 1:06
1:06
what does fda do?
-
 2:28
2:28
what is fda’s role in regulating drugs?
-
 16:18
16:18
how is my medical device classified?
-
 24:00
24:00
is my product a medical device
-
 16:52
16:52
what’s the difference between fda human factors requirements and iec 62366?
-
 49:08
49:08
medical devices 101: an entry level overview of the fda
-
 4:46
4:46
medical device registration in the us- an overview of pathways and classification
-
 57:13
57:13
us postmarket surveillance requirements for medical devices
-
 9:28
9:28
medical device regulations / fda approval